3M Particulate Healthcare Respirator 1860, N95
3M Particulate Healthcare Respirator 1860, N95
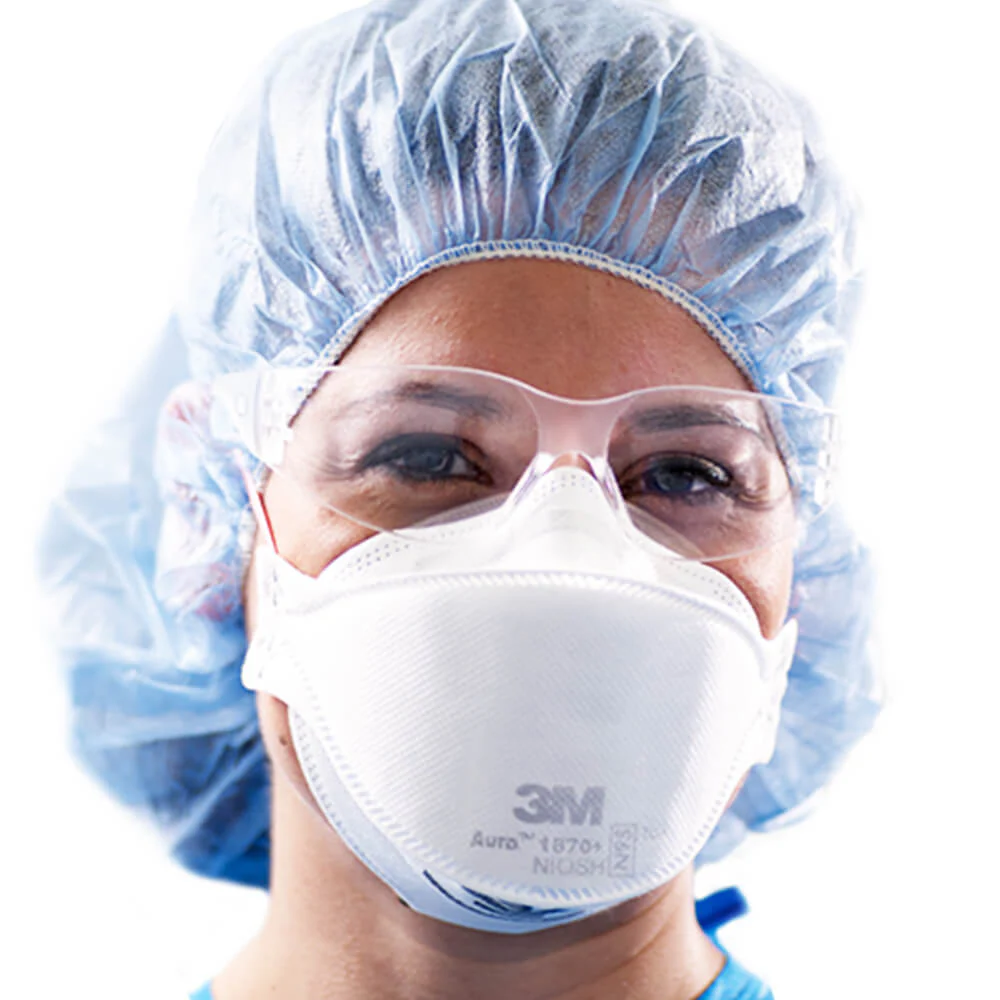
The 3M™ Particulate Healthcare Respirator is designed to help provide respiratory protection for the wearer. It meets CDC guidelines for Mycobacterium tuberculosis exposure control.
As a disposable particulate respirator, it is intended to help reduce wearer exposure to certain airborne particles including those generated by electrocautery, laser surgery, and other powered medical instruments.
As a surgical mask, it is designed to be fluid-resistant to splash and spatter of blood and other infectious materials.
Before use, the wearer must read and understand the User Instructions provided as a part of the product packaging.
Suggested Use:
- Operating Rooms and Clinics
- TB Wards and Infection Control Practices
- Patient Care, Labour and Delivery
- Laboratory, emergency or pandemic preparedness planning
Features:
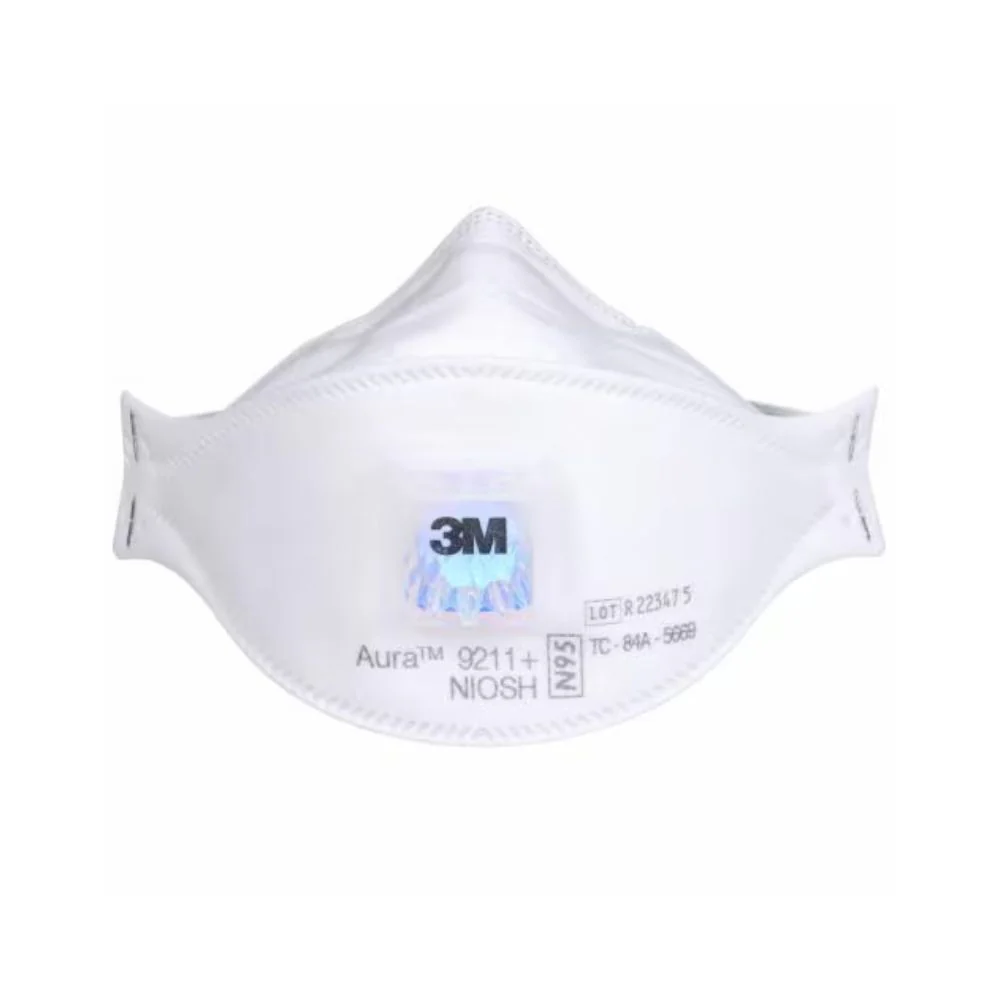
- NIOSH approved N95
- Meets CDC guidelines for Mycobacterium tuberculosis exposure control
- FDA cleared for use as a surgical mask
- >99% Bacterial Filtration Efficiency according to ASTM F2101
- Fluid resistant according to ASTM F1862
- Respirator contains no components made from natural rubber latex

Original: $39.06
-70%$39.06
$11.72Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
3M Particulate Healthcare Respirator 1860, N95
The 3M™ Particulate Healthcare Respirator is designed to help provide respiratory protection for the wearer. It meets CDC guidelines for Mycobacterium tuberculosis exposure control.
As a disposable particulate respirator, it is intended to help reduce wearer exposure to certain airborne particles including those generated by electrocautery, laser surgery, and other powered medical instruments.
As a surgical mask, it is designed to be fluid-resistant to splash and spatter of blood and other infectious materials.
Before use, the wearer must read and understand the User Instructions provided as a part of the product packaging.
Suggested Use:
- Operating Rooms and Clinics
- TB Wards and Infection Control Practices
- Patient Care, Labour and Delivery
- Laboratory, emergency or pandemic preparedness planning
Features:
- NIOSH approved N95
- Meets CDC guidelines for Mycobacterium tuberculosis exposure control
- FDA cleared for use as a surgical mask
- >99% Bacterial Filtration Efficiency according to ASTM F2101
- Fluid resistant according to ASTM F1862
- Respirator contains no components made from natural rubber latex